Monitoring bacteria in the body with ultrasound
January 23, 2018
Monitoring bacteria in the body with ultrasound
At a Glance
- Researchers engineered genes that allowed bacteria in the bodies of mice to be detected with ultrasound.
- With further development, the technique could allow doctors to track bacteria that are sometimes used in treatments for gut infections and cancer.
Trillions of microscopic organisms are living in and on your body every day. Microbes living in your gut help you digest food and play a role in your overall health and well-being. Changes in your gut’s microbes can lead to digestive problems or other health conditions.
Some microbes, such as bacteria, can be used in medical treatments. For instance, taking probiotics (harmless microbes similar to those found in the human gut) may help treat gastrointestinal problems, such as irritable bowel syndrome. Researchers have also investigated using bacteria to deliver drugs to tumors. Being able to track these bacteria within the body could benefit such medical applications.
Current imaging technologies for monitoring gut microbes are limited since light-based methods can’t penetrate deep into the body. To develop a better way to track microorganisms inside the body, a team led by Dr. Mikhail G. Shapiro at California Institute of Technology engineered what they call acoustic reporter genes. These genes enable the detection of bacteria using ultrasound, a high-resolution imaging technique that uses sound waves to detect objects. The research was supported in part by the NIH’s National Institute of Biomedical Imaging and Bioengineering (NIBIB). Results were published in Nature on January 4, 2018.
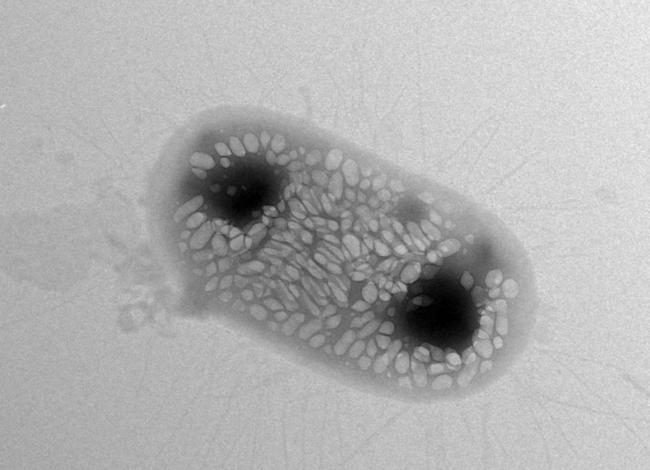
Acoustic reporter genes are genetic constructs that can be inserted into bacteria to direct them to make gas-filled nanostructures called gas vesicles. These gas vesicles can scatter sound waves, making them detectable with ultrasound. Several genes are known to encode the gas-filled vesicles, which normally help water-dwelling bacteria regulate buoyancy.
The researchers took gas-vesicle producing genes from water-dwelling bacteria and inserted them into bacteria known to colonize the gut. E. coli has been used as a probiotic to treat some gut infections. The team found a combination of genes that could make E. coli bacteria produce gas vesicles on their own. This combination of acoustic reporter genes resulted in gas-filled vesicles with the properties required for ultrasound tracking.
The team next looked at whether they could monitor E. coli with the acoustic reporter genes using ultrasound in living mammals. They found that the engineered bacteria, when placed into the gastrointestinal tracts of mice, could be visualized there using ultrasound. They compared the technique with traditional light-based imaging methods that use bacteria containing a light-emitting molecule. The acoustic reporter gene method allowed them to track bacteria deeper in the gut.
The scientists also showed that acoustic reporter genes could be used to monitor S. typhimurium bacteria after they were injected into tumors in mice. This result suggests the technique could potentially aid some cancer treatments.
“We are engineering the bacterial cells so they can bounce sound waves back to us and let us know their location the way a ship or submarine scatters sonar when another ship is looking for it,” says Shapiro. “This is the first acoustic reporter gene for use in ultrasound imaging.”
The researchers hope this technology will revolutionize the imaging of cells within the body. However, it will take years to develop the method for use in humans.
—Tianna Hicklin, Ph.D.
Related Links
- Changing Gut Bacteria in Crohn’s Disease
- Gut Microbes and Diet Interact to Affect Obesity
- Food Additives Alter Gut Microbes, Cause Diseases in Mice
- Diet Affects Autoinflammatory Disease Via Gut Microbes
- The Healthy Human Microbiome
- Keeping Your Gut in Check
- Your Microbes and You
- Human Microbiome Project
References
Acoustic reporter genes for noninvasive imaging of microorganisms in mammalian hosts. Bourdeau RW, Lee-Gosselin A, Lakshmanan A, Farhadi A, Kumar SR, Nety SP, Shapiro MG. Nature. 2018 Jan 3;553(7686):86-90. doi: 10.1038/nature25021. PMID: 29300010.
Funding
NIH’s National Institute of Biomedical Imaging and Bioengineering (NIBIB); Canadian Institute of Health Research; the Pew Charitable Trusts; National Science Foundation; Natural Sciences and Engineering Research Council of Canada; California Institute of Technology; Heritage Medical Research Institute, Burroughs Wellcome Fund; and David and Lucile Packard Foundation.


