Progress toward universal vaccines
Research in Context July 25, 2023
Progress toward universal vaccines
Preparing for the next pandemic before it starts
This article, about the concept of universal vaccines, inaugurates a new periodic feature of NIH Research Matters called Research in Context. These in-depth articles will focus on cutting-edge NIH biomedical research topics, describing the current state of research in a field and where it may be heading.
In March 2020, the world changed in the blink of an eye as countries around the world locked down to slow the spread of COVID-19. This often deadly disease, caused by a previously unknown coronavirus called SARS-CoV-2, seeded outbreaks across the globe in record time. By the end of 2022, it had killed more than 6 million people worldwide.
But COVID-19 was far from humanity’s first pandemic. As recently as 15 years ago, an outbreak of pandemic flu sickened 60 million people globally. In 2003, a new disease called severe acute respiratory syndrome, or SARS, sickened more than 8,000 people worldwide. It was caused by a virus called SARS-associated coronavirus (SARS-CoV). No one can predict when the next pandemic will happen—only that one eventually will.
These recent pandemics have brought into stark relief the need to be prepared for the next emerging disease, whenever it arrives. To this end, NIH-funded research teams have been working to develop universal vaccines against diseases with pandemic potential. Unlike current vaccines, which confer immunity to one or several strains of a disease, universal vaccines are designed to teach the immune system to defend against all versions of a pathogen—even versions that don’t exist yet. They do this by targeting an element of the pathogen that remains the same across all strains and types.
Such targets are usually those that are least accessible to the immune system. This has posed a significant challenge to vaccine researchers. But with recent progress in vaccine technology, researchers believe that universal vaccines are closer to reality than ever before.
Moving beyond educated guesses
For some viruses, the only constant is change. Locked in a continuous battle with the human immune system, many common viruses change, or mutate, rapidly. This means that even if you’ve been infected with a previous version of a virus, your immune system may not recognize an altered version the next time around.
A well-known example of the arms race between viruses and humans is the influenza virus, commonly known as the flu. More than 20 types of the virus—each of which, in turn, contains many different strains—circulate among people and animals, changing almost constantly.
The flu vaccine you get every year targets four strains that the scientific community predicts are most likely to predominate that season. “We have a well-established system to collect [information] on which strains are circulating all over the world,” says NIH vaccine researcher Dr. Karin Bok. “But it takes at least six months from the decision of which [strains] to include to the vaccine being available to the public.” And which flu strains circulate during that time can change unpredictably.
As a result, seasonal flu vaccines vary in their effectiveness. Their ability to prevent severe disease ranges from as high as 60% to as low as 10%.
All widely used flu vaccines to date teach the immune system to recognize a protein called hemagglutinin, which is found on the surface of the influenza virus. The virus uses hemagglutinin to enter human cells.
In a recent NIH-funded study, researchers designed a flu vaccine to provide broad protection against different influenza viruses. To create the vaccine, the researchers fused hemagglutinin to building blocks that assemble into nanometer-sized particles, or nanoparticles. The nanoparticles included hemagglutinin from four different flu strains. The researchers reasoned that this would encourage the immune system to respond to parts of the protein that were more similar, or conserved, between influenza strains.
“What these nanoparticles do is repetitively display the antigen—the protein from the virus—that you’re trying to mount an immune response to,” explains Dr. Neil King of UW Medicine, who helped lead the study along with researchers from NIH’s Vaccine Research Center (VRC). “And repetition…tells the immune system that this is something dangerous.” This approach can create a strong immune memory of the conserved part of these viral proteins.
In studies in mice, ferrets, and monkeys, the nanoparticle vaccines induced antibody responses against the included strains that were as good as or better than those elicited by a commercial vaccine. Notably, the nanoparticle vaccines also provided near-complete protection against several related flu strains that were not included in the nanoparticles. In contrast, the commercial vaccine did not protect against those other strains.
“These nanoparticle vaccines may be what we call a “supra-seasonal vaccine”—a vaccine that protects for more than one year,” King says.
The vaccine, called FluMos-v1, is now in phase 1 clinical trials.
VRC researchers have also been working on another vaccine that may provoke an even broader immune response to influenza. The team based their approach on the structure of hemagglutinin, which consists of a stem and a head. Flu vaccines to date have targeted the head of the protein, which is most accessible to immune cells. But this is also the part of the protein that mutates fastest.
The new VRC vaccine, which has completed an early-phase human trial, uses a nanoparticle to display the hemagglutinin stem without the head. The hemagglutinin stem tends to remain relatively unchanged, even as the head rapidly changes. The trial found that immunization with this vaccine was safe and elicited immune responses to a range of hemagglutinins that lasted more than a year after vaccination.
Showing the immune system the stem has an added advantage, Bok explains: it’s not something the immune system is used to seeing. This novelty provokes a stronger immune response. That, plus the conserved nature of the stem, “may make it so you would be able to mount an immune response to any hemagglutinin you’re exposed to [after vaccination],” Bok says.
A pressing need for universal protection
The nanoparticle technologies pioneered to develop universal flu vaccines are now being tested to create vaccines that could protect against multiple current and future coronaviruses, including SARS-CoV-2.
“SARS-CoV-2 has proven itself capable of making new variants that are prolonging the global COVID-19 pandemic,” says Dr. Pamela Bjorkman, who leads an NIH-funded research team at the California Institute of Technology. And SARS-CoV-2 wasn’t the first virus of its kind—a type called a betacoronavirus—to jump from animals to people. SARS-CoV came before, and so did MERS-CoV, which causes the deadly Middle East Respiratory Syndrome (MERS).
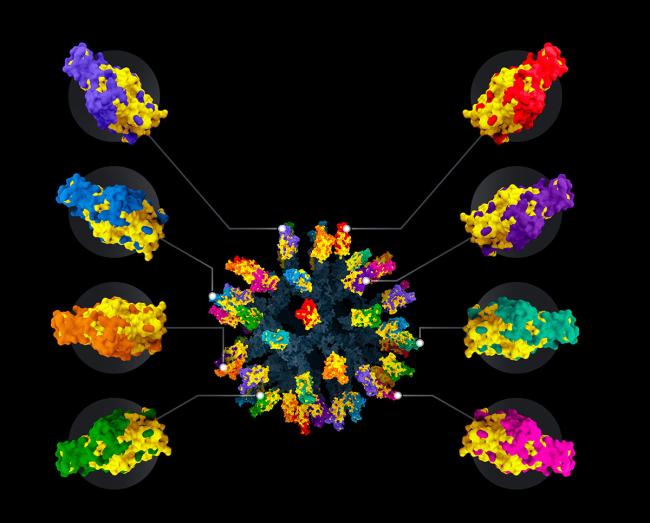
“The fact that three betacoronaviruses—SARS-CoV, MERS-CoV, and SARS-CoV-2—have spilled over into humans from animal hosts in the last 20 years illustrates the need for making broadly protective vaccines,” she says.
In a recent study, Bjorkman and her team combined pieces of the spike proteins from eight different coronaviruses into a new nanoparticle vaccine. The portion of the spike protein they used is called the receptor binding domain, or RBD. Coronaviruses use the RBD to enter human cells.
Each nanoparticle included 60 RBDs, so that any two adjacent ones were rarely from the same coronavirus. As with flu vaccines, this arrangement encourages antibody-producing immune cells to target areas that are similar across the proteins.
The team tested the new vaccine in mice engineered to be vulnerable to SARS-CoV-2. Following vaccination, the mice produced antibodies that recognized a range of different coronaviruses. And as expected, the antibodies recognized parts of the spike protein that remained similar between coronaviruses.
Promising results were also seen when the vaccine was tested in monkeys. The animals were protected not only against a SARS-CoV-2 variant that wasn’t included in the vaccine but also against SARS-CoV.
“We can’t predict which virus or viruses among the vast numbers in animals will evolve in the future to infect humans to cause another epidemic or pandemic,” Bjorkman explains. “What we’re trying to do is make an all-in-one vaccine protective against SARS-like coronaviruses. This sort of vaccine would also protect against current and future SARS-CoV-2 variants without the need for updating.”
The arrival of mRNA vaccines
Another tool being tested to create more broadly effective vaccines is mRNA technology. This technology enabled COVID-19 vaccines to be developed and brought to the clinic within less than a year after the genome of SARS-CoV-2 was sequenced.
Traditionally, vaccines used weakened or killed versions of an actual pathogen, Bok explains. As technology improved, more refined vaccines were made that included only the pathogen proteins that interact with human cells. mRNA vaccines are very similar, Bok says.
“You’re still getting the same protein,” she explains. “It’s just the delivery mechanism that’s different. Instead of giving you the protein, it’s giving [your body] the source code—the software—so you can make [that protein] yourself.”
This approach allows the immune system to be exposed to substantially greater quantities of a protein than with a traditional vaccine. That, in turn, can produce a stronger immune response. Another advantage of mRNA vaccines is that they can be much cheaper to produce and easier to modify quickly. These vaccines are now being tested to prevent a variety of illnesses beyond SARS-CoV-2, including influenza.
An NIH-funded research team led by Dr. Scott Hensley from the University of Pennsylvania designed a vaccine that includes mRNAs for hemagglutinin from all 20 influenza types known to infect people. It hadn’t been possible to include so much variation with traditional vaccine production methods. But the researchers thought it might work with mRNA technology.
In animal tests, mice that received the experimental mRNA vaccine produced antibodies against both similar and unique regions of all 20 different types of hemagglutinin. Levels of these antibodies remained unchanged for months after vaccination. This robust antibody production occurred whether or not the mice had previously been exposed to one of the flu strains.
Further experiments showed that vaccination protected both mice and ferrets from a dangerous flu strain similar to one of those in the vaccine.
“For a conventional vaccine, immunizing against all these types would be a major challenge, but with mRNA technology it’s relatively easy,” Hensley says. “The idea here is to have a vaccine that will give people a baseline level of immune memory to diverse flu strains, so that there will be far less disease and death when the next flu pandemic occurs.”
“When we think about pandemic preparedness,” Bok says, “what we’re most worried about is the first few months, before vaccines can be prepared. Universal vaccines, along with antivirals and other treatments, could provide [vital] protection against severe disease from the next pandemic.”
—by Sharon Reynolds
Related Links
- Bivalent Boosters Provide Better Protection Against Severe COVID-19
- Using mRNA Technology for a Universal Flu Vaccine
- Fighting Current and Future Coronaviruses with a Single Vaccine
- Experimental Vaccine Protects against Multiple Coronaviruses
- Nanoparticle Vaccine against Various Coronaviruses
- Nanoparticle-Based Flu Vaccine
- Universal Mosquito Vaccine Tested
- Newly Identified Flu Antibodies Could Improve Vaccines and Treatments
- Universal Influenza Vaccine Research
- Pandemic Preparedness
References
Quadrivalent influenza nanoparticle vaccines induce broad protection. Boyoglu-Barnum S, Ellis D, Gillespie RA, Hutchinson GB, Park YJ, Moin SM, Acton OJ, Ravichandran R, Murphy M, Pettie D, Matheson N, Carter L, Creanga A, Watson MJ, Kephart S, Ataca S, Vaile JR, Ueda G, Crank MC, Stewart L, Lee KK, Guttman M, Baker D, Mascola JR, Veesler D, Graham BS, King NP, Kanekiyo M. Nature. 2021 Apr;592(7855):623-628. doi: 10.1038/s41586-021-03365-x. Epub 2021 Mar 24. PMID: 33762730.
A multivalent nucleoside-modified mRNA vaccine against all known influenza virus subtypes. Arevalo CP, Bolton MJ, Le Sage V, Ye N, Furey C, Muramatsu H, Alameh MG, Pardi N, Drapeau EM, Parkhouse K, Garretson T, Morris JS, Moncla LH, Tam YK, Fan SHY, Lakdawala SS, Weissman D, Hensley SE. Science. 2022 Nov 25;378(6622):899-904. doi: 10.1126/science.abm0271. Epub 2022 Nov 24. PMID: 36423275.
Mosaic RBD nanoparticles protect against challenge by diverse sarbecoviruses in animal models. Cohen AA, van Doremalen N, Greaney AJ, Andersen H, Sharma A, Starr TN, Keeffe JR, Fan C, Schulz JE, Gnanapragasam PNP, Kakutani LM, West AP Jr, Saturday G, Lee YE, Gao H, Jette CA, Lewis MG, Tan TK, Townsend AR, Bloom JD, Munster VJ, Bjorkman PJ. Science. 2022 Aug 5;377(6606):eabq0839. doi: 10.1126/science.abq0839. Epub 2022 Aug 5. PMID: 35857620.
An influenza hemagglutinin stem nanoparticle vaccine induces cross-group 1 neutralizing antibodies in healthy adults. Widge AT, Hofstetter AR, Houser KV, Awan SF, Chen GL, Burgos Florez MC, Berkowitz NM, Mendoza F, Hendel CS, Holman LA, Gordon IJ, Apte P, Liang CJ, Gaudinski MR, Coates EE, Strom L, Wycuff D, Vazquez S, Stein JA, Gall JG, Adams WC, Carlton K, Gillespie RA, Creanga A, Crank MC, Andrews SF, Castro M, Serebryannyy LA, Narpala SR, Hatcher C, Lin BC, O'Connell S, Freyn AW, Rosado VC, Nachbagauer R, Palese P, Kanekiyo M, McDermott AB, Koup RA, Dropulic LK, Graham BS, Mascola JR, Ledgerwood JE; VRC 321 study team. Sci Transl Med. 2023 Apr 19;15(692):eade4790. doi: 10.1126/scitranslmed.ade4790. Epub 2023 Apr 19. PMID: 37075129.
An influenza H1 hemagglutinin stem-only immunogen elicits a broadly cross-reactive B cell response in humans. Andrews SF, Cominsky LY, Shimberg GD, Gillespie RA, Gorman J, Raab JE, Brand J, Creanga A, Gajjala SR, Narpala S, Cheung CSF, Harris DR, Zhou T, Gordon I, Holman L, Mendoza F, Houser KV, Chen GL, Mascola JR, Graham BS, Kwong PD, Widge A, Dropulic LK, Ledgerwood JE, Kanekiyo M, McDermott AB. Sci Transl Med. 2023 Apr 19;15(692):eade4976. doi: 10.1126/scitranslmed.ade4976. Epub 2023 Apr 19. PMID: 37075126.


