Universal mosquito vaccine tested
June 23, 2020
Universal mosquito vaccine tested
At a Glance
- A small trial showed that a vaccine designed to provide broad protection against mosquito-borne diseases is safe and causes a strong immune response in healthy volunteers.
- More studies are needed to test its effectiveness against specific diseases.
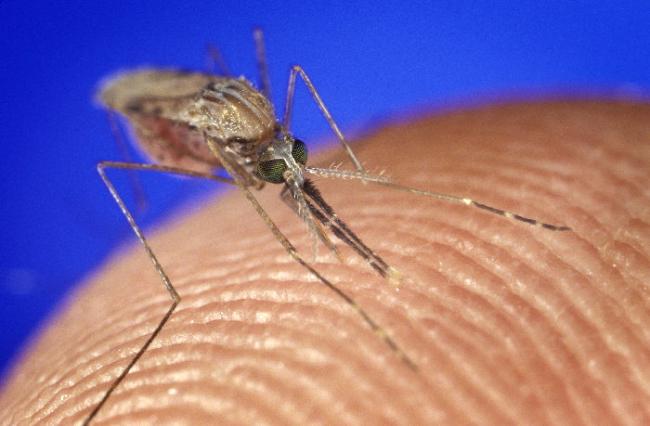
Most mosquito bites are harmless. But some mosquitoes carry pathogens, like bacteria and viruses, that can be deadly. Mosquito-related illnesses kill hundreds of thousands of people worldwide each year.
When a mosquito bites you, compounds in its saliva interact with your body’s initial immune response to help pathogens evade your body’s disease defenses. Studies have shown that animals that have antibodies to mosquito saliva proteins have some protection against mosquito-borne diseases. Although there are vaccines being studied for individual mosquito-borne diseases—like malaria, West Nile virus, and dengue fever—a vaccine against mosquito saliva could provide broader protection against many diseases.
To investigate, a research team led by Drs. Jessica Manning and Matthew Memoli at NIH’s National Institute of Allergy and Infectious Diseases (NIAID) tested a “universal mosquito vaccine” known as AGS-v. It contains four synthetic peptides that are found in the salivary gland proteins of different mosquito species. The work was funded by NIAID along with NIH’s Office of the Director (OD) and National Cancer Institute (NCI). Results were published on June 11, 2020 in the Lancet.
Forty-nine healthy volunteers were randomly assigned to one of three groups. One group received injections of an inactive placebo (sterile water); one received injections of the vaccine; and one received injections of the vaccine combined with an adjuvant, a compound designed to enhance immune responses. They were given two injections 21 days apart.
On the 42nd day of the study, volunteers underwent a “feeding session” with disease-free mosquitoes bred at the NIH. The mosquitoes fed briefly from volunteers’ arms through a fine mesh while in a container resembling a cup.
The volunteers were monitored for twelve months after the first vaccination. None had serious adverse reactions to the vaccine or mosquito feeding. The mosquitoes were also monitored after the feeding session to see whether the vaccine had any impact on their survival or number of offspring they produced.
The volunteers’ blood tests showed that those who received the vaccine with adjuvant produced specific antibodies to the peptides. One of the volunteers developed a significant rash after the first dose of the vaccine, but no larger safety concerns were identified. The mosquitoes didn’t seem to be affected after feeding on people who’d been vaccinated.
“A widely available ‘universal’ vaccine could possibly provide protection against emerging and re-emerging mosquito-borne diseases as they arise, allowing public health officials to quickly respond to new outbreaks and epidemics without waiting for new treatments or vaccines to be developed,” Manning says.
More research is needed to test whether the vaccine would work against individual pathogens and with exposure to mosquitoes in a natural environment.
Related Links
- Engineering Malaria Resistance in Mosquitoes
- Progress on Malaria Vaccines
- Experimental Malaria Vaccine Protects Adults for More Than a Year
- Malaria Vaccine Found Safe and Protective
- Experimental Vaccine Protects Against Dengue Virus
- How Dengue Virus Infects Cells
- DNA Vaccines Protect Monkeys Against Zika Virus
- Block the Buzzing, Bites, and Bumps: Preventing Mosquito-Borne Illnesses
- From A to Zika: Understanding Emerging Diseases
- West Nile Virus
- Dengue Fever
- Malaria
- Zika Virus Infection
- Zika Virus Information and Resources


