How cancer vesicles breach the blood-brain barrier
October 1, 2019
How cancer vesicles breach the blood-brain barrier
At a Glance.
- Researchers discovered how small particles from cancer cells called extracellular vesicles cross the blood-brain barrier to make the brain more hospitable to metastatic tumors.
- A detailed understanding of this process could guide strategies to stop brain metastases as well as methods to deliver drugs to the brain.
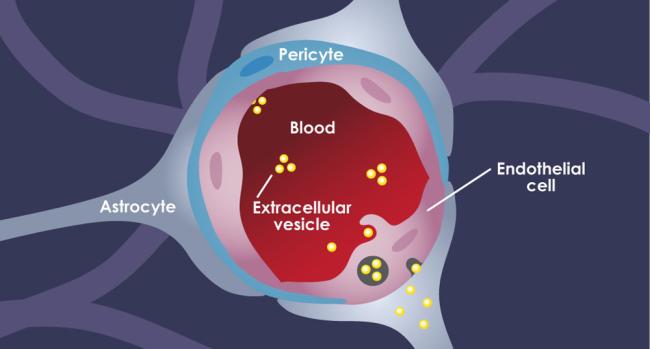
The brain is walled off from the rest of the body by the blood-brain barrier. This tightly packed mix of cells sits between the blood vessels that lead to the brain and the brain tissue itself. The blood-brain barrier helps protect the brain from threats like infection. But it can also stop helpful medications from getting to the brain when needed.
Researchers know that the blood-brain barrier isn’t perfect. For example, cancer cells can sometimes get past it and establish metastatic tumors (ones from other locations) in the brain. If scientists could better understand how cancer cells accomplish this crossing, they might be able to develop methods to prevent it.
Many cells, including cancer cells, release tiny sacs called extracellular vesicles (EVs). EVs can affect other cells by transferring proteins and genetic material into them. EVs from tumors, for example, can enter the circulation and alter distant organs to make them more susceptible to metastatic cancer. Because of their ability to alter cells, EVs are being studied as potential therapeutics for cancer and other diseases.
A research team led by Drs. Marsha Moses and Golnaz Morad from Boston Children’s Hospital and Harvard Medical School investigated whether EVs can cross the blood-brain barrier and play a role in brain metastases. The research was funded in part by NIH’s National Cancer Institute (NCI). Results were published on September 3, 2019, in ACS Nano.
The team first injected mice with EVs released from breast cancer cells known to form tumors in the brain. Mice injected with EVs from these cells developed more and larger brain metastases than mice that received EVs from breast cancer cells that hadn’t turned metastatic. Fluorescent tagging of the EVs showed that they crossed the blood-brain barrier.
The researchers next used their laboratory models of the blood-brain barrier to find out how the EVs crossed into the brain. They found that EVs were able to cross the cells of the blood-brain barrier through a process called transcytosis. They were first engulfed with other materials, then moved through the cell, and finally fused with the cell membrane on the other side to be released. In this way, their contents could pass through the blood-brain barrier to the brain.
Using fluorescent imaging of live zebrafish embryos, the researchers were able to watch EVs travel through cells and fuse with cell membranes to be released on the other side.
The team also showed that EVs from metastatic cancer cells altered blood-brain barrier cells to disrupt their ability to break down vesicles. This would allow more EVs to make their way safely across the barrier cells and into the brain. Once inside, the EVs help make the region more hospitable to metastatic cancer cells.
“EVs can manipulate endothelial cells to facilitate their own transport across the blood-brain barrier,” Morad says. “They hijack the pathways involved in the uptake and sorting of molecules.”
The team is now testing whether EVs could be engineered to deliver anticancer drugs across the blood-brain barrier.
—by Sharon Reynolds
Related Links
- Blood-Brain Barrier Test May Predict Dementia
- Most Tumors in Body Share Important Mutations
- Novel Approach Gives Insights Into Tumor Development
- Genes Help Breast Cancer Cells Invade the Brain
- Metastatic Cancer
- Metastatic Cancer Research
References
Tumor-Derived Extracellular Vesicles Breach the Intact Blood-Brain Barrier via Transcytosis. Morad G, Carman CV, Hagedorn EJ, Perlin JR, Zon LI, Mustafaoglu N, Park TE, Ingber DE, Daisy CC, Moses MA. ACS Nano. 2019 Sep 10. doi: 10.1021/acsnano.9b04397. [Epub ahead of print]. PMID: 31479239.
Funding
NIH’s National Cancer Institute (NCI) and National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK); Breast Cancer Research Foundation; Advanced Medical Foundation.


