Extending preservation time for donated livers
September 24, 2019
Extending preservation time for donated livers
At a Glance
- A new preservation method protected human livers during cooling and kept them healthy for more than 24 hours.
- Prolonging the storage life of donated organs could eventually help increase the number of potential transplants.
Every year, there are not enough donor livers available for the people who need liver transplants. This mismatch is not only caused by a shortage of organ donors. It can also sometimes be hard to deliver a donated liver to the person who needs it.
With existing techniques, human organs can be preserved for an average of nine hours. Depending on where a donor and recipient live, this may not be enough time to transport the organ and prepare for surgery.
Researchers led by Dr. Korkut Uygun of Harvard Medical School have been working on a system to extend the storage time of donated organs. In previous work, they developed a procedure called supercooling that could successfully preserve rat livers at below-freezing temperatures for several days.
However, the technique did not work in human livers, which are about 200 times larger than rat livers. Their large size makes human livers vulnerable to ice formation during cooling. When ice forms in organs, it can cause irreversible tissue damage.
In a new study, Dr. Uygun and his colleagues made several modifications to their cold storage technique to eliminate ice formation. They tested the new technique in human livers that had been rejected from a transplant program. The work was supported in part by NIH’s National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), National Institute of Biomedical Imaging and Bioengineering (NIBIB), and National Heart, Lung, and Blood Institute (NHLBI). Results were published on September 9, 2019, in Nature Biotechnology.
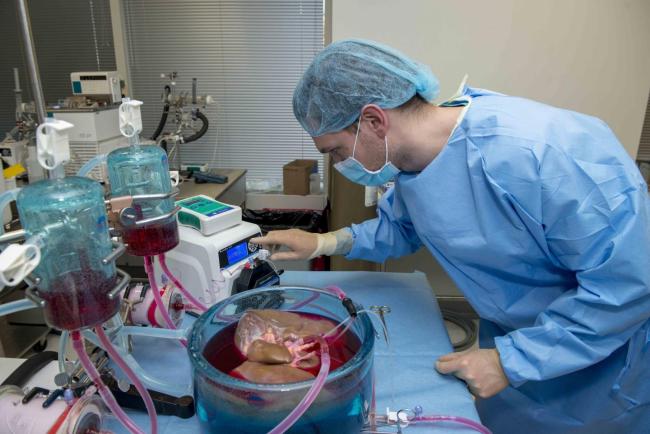
First, the researchers removed all air from the storage system before preservation. Air can promote ice crystal formation and growth. Second, they added two additional chemicals to their cocktail of protective agents. Finally, they used a machine to slowly and evenly push the protective solution through the liver tissue instead of performing this step by hand.
The team measured markers of liver health, including energy usage and bile production, before and after the storage of five livers. These measurements remained similar for each liver from before storage to after re-warming, indicating that they remained undamaged.
The team next re-warmed an additional three supercooled livers and flowed blood products through them to mimic what they would undergo when transplanted into a body. Again, the measurements of liver health remained positive. Samples of liver tissue looked healthy when observed under a microscope.
“Delivering viable organs to matching recipients within the window of viability can often be the most challenging aspect of organ transplantation,” says Dr. Seila Selimovic, director of NIBIB's Engineered Tissues program. “By giving doctors and patients more time, this research could someday affect thousands of patients who are waiting for liver or other organ transplants.”
Additional research is needed before the new technique can be tested in the clinic. This includes monitoring long-term survival of preserved livers after transplantation in a large animal model.
Related Links
- Preserving Livers for Transplantation
- Liver Transplant
- Organ Donation: Pass it On
- Fixing Flawed Body Parts: Engineering New Tissues and Organs
References
Supercooling extends preservation time of human livers. de Vries RJ, Tessier SN, Banik PD, Nagpal S, Cronin SEJ, Ozer S, Hafiz EOA, van Gulik TM, Yarmush ML, Markmann JF, Toner M, Yeh H, Uygun K. Nat Biotechnol. 2019 Sep 9. doi: 10.1038/s41587-019-0223-y. [Epub ahead of print] PMID: 31501557.
Funding
NIH’s National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), National Institute of Biomedical Imaging and Bioengineering (NIBIB), and National Heart, Lung, and Blood Institute (NHLBI); U.S. Department of Defense; U.S. Army Medical Research and Development Command; Michael van Vloten Fund for Surgical Research.


